4.7
Full-service medical product design, engineering, and manufacturing.
Need Help Bringing Your Medical Device to Market?
We help doctors, dentists, and companies develop innovative medical devices






Leverage Our Experience in Your Medical Device Category

Surgical Instruments
Turn your ideas for better surgical tools and instruments into working prototypes, ready for manufacturing.

Drug Delivery Devices
Realize your product visions for innovative drug delivery devices that empower medical professionals and patients.

Assistive Devices
Modern technology is creating new opportunities and assistive device product ideas to help people with disabilities.

Vital Sign Monitoring
Sensor technology is fueling the rise of innovative vital sign devices, making new products and ideas possible.
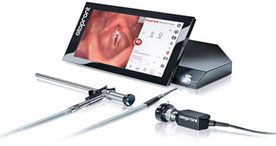
Endoscope 3D Imaging
Endoscope technology continues to get smaller and more powerful, aided by compact chipsets and cameras.
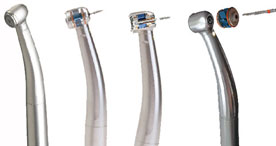
Dental Equipment
Transform your dental device vision into a full-functional prototype for user testing, and full volume production.

Wearable Health Devices
Wearable devices have moved away from novelty to medical use. Start moving your wearable device idea to market.

Biotech Lab Equipment and Instruments
Advances in BioTech lab equipment are bringing innovative product ideas to market faster than ever.

AI/ML Clinical Intelligence Devices
The rise of generative AI and machine learning is helping medical devices become more effective and accurate.
Leverage Our Experience in Your Medical Device Category

Neuromodulation Pain Therapy System

FDA Approved Pelvic Floor Rejuvenation

RFID Pediatric Protection Bracelet

Connected Vital Sign Monitor

Virtual Health Monitoring Device

Revolutionary Dental Drill System
- 27 Years Experience
- 75+ Design Awards
- 1,000+ Manufactured Products
Proven 4-Step Medical Device Product Development Process

Concept Design
Turn your medical device concept into a refined design, assess risk factors, and create architecture plans ready for engineering.
Detailed Engineering
Iterate on design features and create full regulator documentation for a pre-production prototype, ready for clinical trials.

Prototype and Test
Validate product requirements, confirm usability, secure suppliers, and create a fully functioning prototype for user feedback
Manufacturing Setup and Regulatory Approval
Select the most reliable manufacturer, implement quality-control processes, apply for regulatory approval and oversee production — turning your medical device idea into reality.

Frequently Asked Questions
Depending on your new product design’s complexity, it will take 2 to 12 months to complete the 4-stage development to the manufacturing process. Your design idea goes through concept exploration and engineering, 3D prototyping, user testing, and transfer to manufacturing.
Once the design is well documented, manufacturers can bid on it. Our creative team will find a solution that differentiates your product and delivers an ‘I love it’ user response aligned with a product cost that meets business needs, and the manufacturers want to produce.
Need help with your first pilot product build?
We can navigate, negotiate, document, and fine-tune product quality during the initial production batch builds and on to continuous production. Our process ensures we bring value and transition out of the project as your operations team takes over production management. We can continue to facilitate production if your business objectives are to be a more virtual, low-overhead operation.
Our manufacturing setup experts will help you identify and secure trusted part suppliers and a contract manufacturer (CM) onshore, or offshore, depending on your product objectives and needs.
We can facilitate the prototyping and product builds, from helping coordinate the CM and parts supplier relationships, to overseeing product batch builds, injection tooling approvals, release controlled documentation, quality plans, regulatory testing, Bill of Materials managements, and parts supply coordination.
Depending on your medical device requirements we can support with regulatory approvals including FDA and Health Canada.
Here are reasons why our client’s choose us:
- We have all the product development expertise you need in-house including: design, engineering, software, and manufacturing
- Our senior regulatory expert will directly handle FDA and Health Canada approvals, speeding up the process
- You’ll benefit from faster market entry, onsite prototype creation, and access to a global network of suppliers and manufacturers.
- We can share successful client stories in medical device development to illustrate our effectiveness. Check-out our most recent success: Joylux
NDA’s ensure confidentiality for both parties during conversations. We provide a mutual NDA where the terms apply to both parties. If your organization has an NDA that requires our signature, we handle this through email.
Designing a new hardware product starts with a Concept Design (CD) stage. First, business, marketing, and design teams plan a potential product concept to deliver unique customer value.
An experienced development team can facilitate the requirements definition, oversee the creative design engineering process, provide manufacturing feasibility, and estimate the cost of product and development. A concept design exploration and product architecture definition for a new product idea takes 200 or more hours, depending on the knowledge of the user-buyer, the complexity of the design, the number of custom components, product safety requirements, and any formal documentation required.
Expert fully integrated product Design/Engineering teams in North America have rates from $100 to $ 400 per hour. After a phone discussion, we provide a Design budget based on your project’s specific needs. Call us for a chat.
From medical device concept to volume manufacturing. We help you get there.
Design 1st integrates our knowledge and expertise with yours to bring your product idea to life
Have a product that needs building?
what happens next
- We review your brief
- You receive an NDA + D1 overview
- 30-minute intro call
- Development plan with timeline and budget
Prefer to talk?
Tell Us About Your Project
We reply within 1 business day with an NDA and a next step.
"(Required)" indicates required fields
what happens next
- We review your brief
- You receive an NDA + D1 overview
- 30-minute intro call
- Development plan with timeline and budget




