For Medical Devices That Need to Get Built
You have a device idea.
We've built 35+ of them.
We help medical professionals and companies develop innovative medical devices






We work in your specialty
Experience across every medical device category.
If you’re a physician with an idea in one of these areas, we’ve built devices like yours before.

Surgical Instruments
Turn your ideas for better surgical tools and instruments into working prototypes, ready for manufacturing.

Drug Delivery Devices
Realize your product visions for innovative drug delivery devices that empower medical professionals and patients.

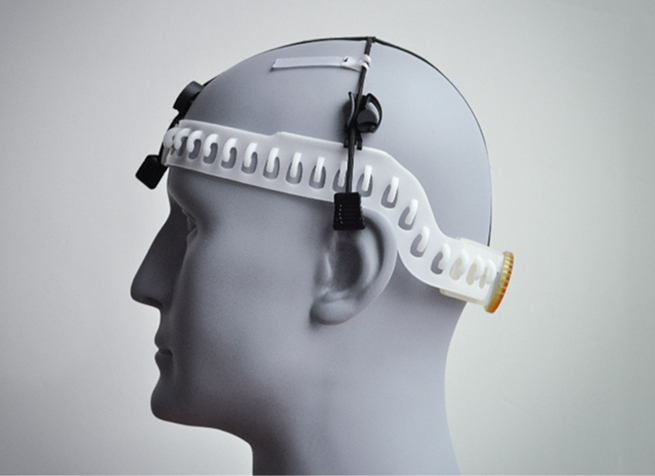
Assistive Devices
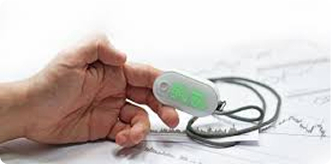
Vital Sign Monitoring
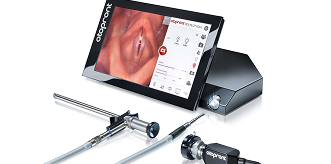
Endoscope & Imaging

Dental Equipment

Wearable Health Devices

Biotech Lab Equipment

AI/ML Clinical Devices
A Product Development Company With the Experience You Need
25+
Physician-invented
devices developed
16 yr
Medical device
experience
100%
IP retained
by clients
$0
Equity taken.
Ever.
The 5 questions every physician asks
If you have a device idea, you've already asked yourself these.
There’s no universal first step. There’s only YOUR first step — based on where you are, what you have, and where the real risks are.
Some doctors come in with a napkin sketch. Some come in with a working prototype. Some have a patent already filed. The starting point is different every time.
We assess where you actually are, then identify the 2-3 decisions that matter most right now. Not a 47-step checklist. The next 2-3 things.
Sometimes the answer is: stop. A fatal flaw, a regulatory barrier, an existing patent. That’s valuable information that saves you $50K and two years.
You don’t need to become an engineer. Stay a physician. Your clinical insight IS the value.
How it works: 30-60 minute calls, weekly or bi-weekly. You make the decisions. We execute between calls. Flexible scheduling that works around your practice, your patients, your life.
You decide what the device should DO. We figure out how to BUILD it. Every physician client we’ve worked with kept practicing medicine throughout development. 25+ and counting.
FDA is navigable. It’s not magic — it’s process. Most physician-invented devices are Class I or Class II with established pathways.
Realistic timelines: 6-12 months for design and prototyping. 3-6 months for FDA review. 9-18 months total from concept to manufacturing-ready.
The critical thing: understand your regulatory pathway BEFORE you spend money on prototyping. The pathway determines design constraints, documentation, testing, and cost.
That question has a real answer. We offer a free initial project assessment — no charge, no commitment. The explicit purpose is to tell you the hard truths early, before you spend a dime.
Common mistakes we catch: $30K on a patent before knowing if the device can be manufactured. Prototypes that work on the bench but fall apart in an autoclave. Designs where the tooling cost kills the business case.
Also: does your employment contract assign inventions to your employer? We ask this in the first conversation. If your hospital owns your idea, nothing else matters until that’s resolved.
We don’t take equity. We don’t take ownership. We don’t take revenue share. You hire us at an hourly rate and you keep 100% of your IP.
Phased development with clear milestones. Each phase has a defined scope and cost. At the end of each phase, you decide: continue, pause, or stop. No penalty for stopping.
Realistic cost ranges stated upfront: Class I ($150K-$250K), Class II 510(k) ($250K-$500K+), complex electromechanical ($500K-$1M+). We tell you this before you start, not after.
We work in your specialty
These started as ideas too.
If you’re a physician with an idea in one of these areas, we’ve built devices like yours before.
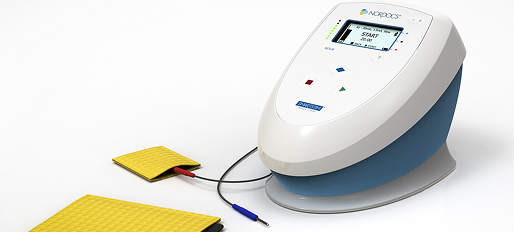



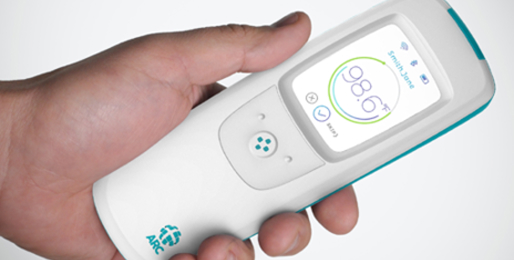
Arc / Lenovo Vital Signs
Stanley PedZ
What we tell you that others won't
The hard truths most development firms skip.
Medical devices cost more than you think.
Sometimes we tell you to stop.
Your hospital might own your idea.
We don't take equity. Ever.
Ready to find out where you stand?
Book a Free Project Review
30 minutes with our senior technical team. We tell you exactly where you stand — what’s viable, what the risks are, and what your next 2–3 decisions should be. No pitch. No commitment.